Sarcoplasmic Reticulum Function in Animal Muscle Cells
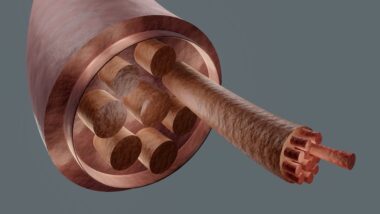
The sarcoplasmic reticulum (SR) is an essential organelle within muscle cells that plays a critical role in muscle contraction and relaxation. This specialized form of the endoplasmic reticulum is found in the cytoplasm of myocytes (muscle cells), primarily responsible for regulating calcium ion concentrations. Its primary function is to store calcium ions while the muscle is at rest, and release them during muscle contraction. When an action potential arrives, it triggers the SR to release calcium ions into the cytoplasm, leading to the activation of contraction mechanisms within the muscle fibers. The calcium-binding protein, calsequestrin, plays a pivotal role here, binding calcium ions at high concentrations within the SR, thereby facilitating efficient calcium storage and release. The SR is not uniform; it consists of different regions, including terminal cisternae, which are specialized for calcium storage adjacent to the transverse tubules. These structural adaptations ensure rapid calcium signaling, crucial for the synchronous contraction of muscle fibers. Dysfunction in the SR can result in muscular diseases, emphasizing its importance in muscle physiology. Overall, the sarcoplasmic reticulum’s intricate structure and function is vital for muscle function.
Calcium Signaling and Muscle Contraction
Calcium signaling within the muscle cells begins with the release of calcium from the sarcoplasmic reticulum into the cytosol, where it binds to troponin on the thin filaments. This binding causes a conformational change in the troponin-tropomyosin complex, ultimately exposing the myosin-binding sites on the actin filaments. Myosin heads, which are part of the thick filaments, then attach to the exposed binding sites, leading to the cross-bridge cycle—a series of interactions that result in muscle contraction. The cyclical process involves ATP hydrolysis and creates force generation through the swinging motion of myosin heads. When calcium is reabsorbed back into the sarcoplasmic reticulum, troponin again changes shape, covering the binding sites and leading to muscle relaxation. This tightly regulated release and reuptake of calcium ions is essential for muscle functionality. The role of various calcium channels, such as Ryanodine Receptors (RyR) and voltage-dependent calcium channels, also underline the complexities of calcium dynamics in muscle cells. Proper calcium handling is critical for maintaining overall muscle health and function.
Further contributions to the understanding of the sarcoplasmic reticulum’s function have emerged through studies in both smooth and striated muscle types. In striated muscles like skeletal and cardiac muscle, the sarcoplasmic reticulum’s organization and function are well characterized. However, smooth muscle exhibits a less extensive SR network, relying more on extracellular calcium influx. In contrast, striated muscle relies on a well-developed network of the sarcoplasmic reticulum that functions in concert with transverse tubules to facilitate rapid calcium signaling. The effectiveness of this system is evident in the synchronous contraction of cardiac muscle, where erratic calcium release can lead to arrhythmias. Furthermore, adaptations in the sarcoplasmic reticulum are also observed in response to various physiological conditions and exercise. Endurance training, for instance, can lead to enhanced calcium handling and improved muscle performance. These adaptations exemplify the plasticity and adaptability of muscle tissue in response to external stimuli. With the understanding of calcium signaling and its dependence on an intact sarcoplasmic reticulum, researchers continue to explore potential therapeutic targets for muscle-related diseases.
Pathologies Associated with Sarcoplasmic Reticulum Dysfunction
When discussing muscle physiology, it is vital to stress the possible pathologies linked to sarcoplasmic reticulum dysfunction. Disease states, such as muscular dystrophies and various cardiomyopathies, can often trace their roots back to defects in sarcoplasmic reticulum function. For example, Duchenne Muscular Dystrophy leads to abnormal calcium handling within the muscle cells. Such alterations can precipitate cycles of damage and degeneration in muscle tissue due to unregulated calcium influx. Similarly, certain types of cardiomyopathies are also associated with mutations in proteins connected to the sarcoplasmic reticulum, also leading to disturbed calcium homeostasis. Research on these diseases emphasizes the critical nature of the SR in muscle function and preservation. Moreover, alterations in calcium signaling are not restricted to inherited diseases; they can also occur as a result of aging, where muscle fibers display impaired calcium handling. Evidence supports that age-related changes in the sarcoplasmic reticulum significantly contribute to reduced muscle force and functionality. Investigating these pathologies highlights the importance of the sarcoplasmic reticulum in both health and disease, as it plays a central role in maintaining muscle integrity.
Advancements in imaging and molecular biology have illuminated the nuances of sarcoplasmic reticulum function. Innovative imaging techniques, such as fluorescence microscopy and electron microscopy, facilitate real-time observation of calcium dynamics within muscle cells. Researchers utilize genetically encoded calcium indicators to track cytosolic calcium fluctuations during muscle contractions, which provides insights into SR-related signaling processes. Additionally, assays for measuring calcium release from the sarcoplasmic reticulum have grown increasingly sophisticated, contributing to a deeper understanding of the molecular pathways involved. Such techniques are crucial for assessing the impact of pharmaceuticals on calcium metabolism and muscle contraction. A greater understanding of SR function also paves the way for developing targeted gene therapies aimed at correcting calcium dysregulation in various myopathies. Indeed, scientists are exploring the use of molecular chaperones and buffering agents as potential treatments for restoring normal calcium handling in damaged muscle cells. Consequently, continued research into sarcoplasmic reticulum physiology, alongside cutting-edge technology, promises novel therapeutic approaches for a variety of muscle-related diseases.
Conclusion: The Importance of Continuous Research
In conclusion, the sarcoplasmic reticulum’s role in animal muscle cells cannot be overstated. It serves as a critical hub for calcium signaling, subsequently influencing muscle contraction and relaxation. Understanding the intricacies of the SR is not only essential from a physiological perspective but also crucial for therapeutic development in muscle pathologies. As research delves deeper into the specific contributions of the sarcoplasmic reticulum, new insights continue to emerge regarding its function and regulation. Advancements in technology and molecular biology will undoubtedly enhance our understanding and may reveal novel intervention strategies to combat muscle diseases. Researchers remain optimistic about the potential for discovering drugs targeting calcium handling in muscle cells, significantly improving conditions for individuals affected by muscle dysfunction. Enhanced awareness of how the SR operates under various conditions—such as exercise, disease, and aging—will further contribute to developing personalized medical treatments. Thus, the field must prioritize ongoing research, ensuring a comprehensive understanding of the sarcoplasmic reticulum’s influence on muscle cells, with the aim of improving health outcomes related to muscle diseases.
Ultimately, the functionality of the sarcoplasmic reticulum in animal muscle cells is a complex interplay of molecular, cellular, and physiological processes. It highlights the need for multidisciplinary approaches to tackle the underlying mechanisms that govern calcium dynamics. Its critical function extends beyond merely sequestering and releasing calcium ions; it involves intricate regulatory pathways that respond to various stimuli, feedback mechanisms, and adaptations to stressors. As research continues to expand our knowledge of the sarcoplasmic reticulum and its role in muscle physiology, it also opens doors for potential therapies that can ameliorate conditions arising from calcium homeostasis disturbances. With promising developments on the horizon, the future of research in this field appears bright. Innovations in therapeutic approaches could provide relief to millions suffering from muscle diseases related to sarcoplasmic reticulum dysfunction. Additionally, interdisciplinary collaboration is vital in this journey—integrating expertise from biology, physiology, and medicine into a unified quest for solutions. Ultimately, the continued investigation of the sarcoplasmic reticulum functions will not just enhance academic knowledge but could translate into significant improvements in clinical practice and patient care.